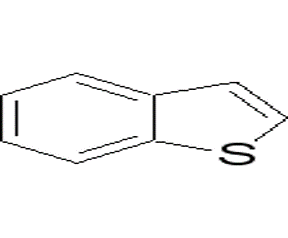
Thianaphthene CAS 95-15-8
Introduction
Toxin peptides are a class of toxic peptide molecules, usually produced by bacteria, plants, or animals. The properties of toxin peptides depend on their structure and source, and some are neurotoxic, cytotoxic, or otherwise biological. CALCISEPTINE IS A TOXIN PEPTIDE WITH CAS NUMBER 178805-91-9, AND ITS SPECIFIC PROPERTIES REQUIRE FURTHER INVESTIGATION.
The preparation of toxin peptides usually needs to be extracted from living organisms or obtained by synthetic methods, and the preparation process can be complex and requires professional technical support.
Toxin peptides are potentially toxic, and the dosage and concentration need to be strictly controlled when used to avoid harm to the human body or the environment. When operating in the laboratory, you need to wear personal protective equipment and follow the relevant safety operating procedures. For the research and application of toxin peptides, relevant regulations and safety standards should be followed to ensure the safety of people and the environment.